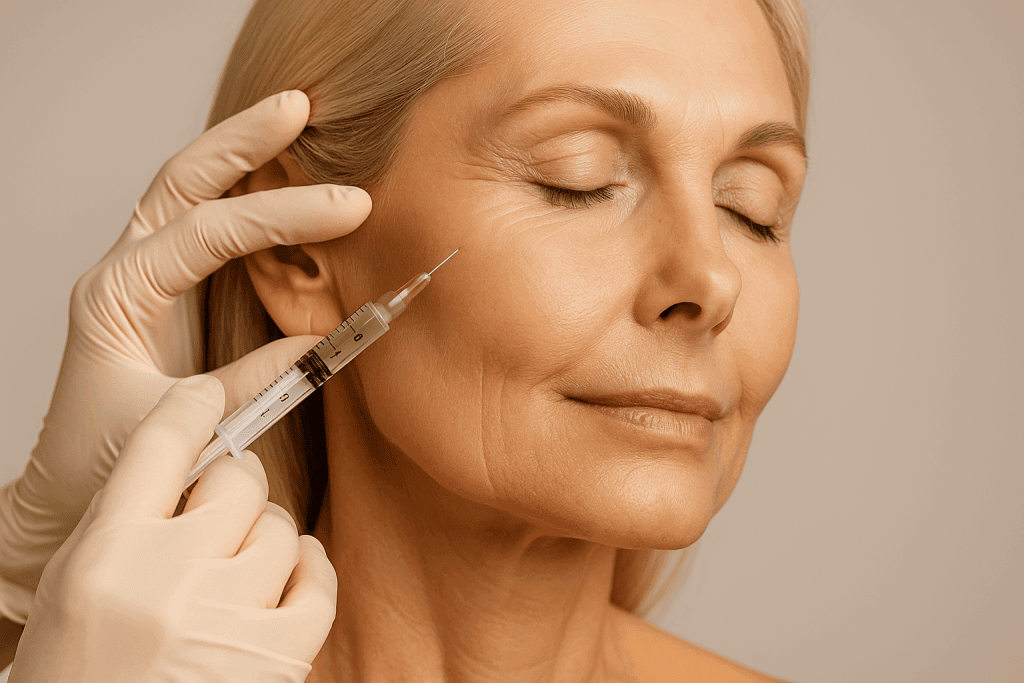
The world of facial aesthetics and reconstruction is a field of medicine that continues to evolve along with the discovery of new therapies and drugs. One form of therapy currently being widely researched is stem cell-based therapy. This therapy has been implemented for skin and muscle tissue regeneration, facial rejuvenation, and other aesthetic procedures.
Research and development of stem cell therapy in the field of facial aesthetics and reconstruction is currently focused on the development of therapies that use stem cells.adipose tissue and umbilical cord tissue, and the integration of stem cells with other clinical procedures. This integration includes fat transfer techniques.for added volume and action microneedling commonly used in the aesthetic sphere to improve skin texture and quality [1].
In general, stem cells operate through complex mechanisms. These cells have the ability to differentiate into various other cell types, as well as act as immunomodulators and catalysts for tissue repair through paracrine mechanisms. [2]. In plastic surgery practice, the clinical application of mesenchymal stem cells ishas shown good results in several indications, including:
-
- wound healing enhance healing that occurs in the wound area by releasing certain molecules (cytokines, growth factorand exosome [3].
- Scar: improve the quality of scar tissue and hypertrophic scars [4]
- Skin rejuvenation and aesthetics: baldness management and increasing the survival rate oftissue lume in facial reconstruction [5].
Currently, stem cell therapy services in reconstructive and aesthetic plastic surgery cases can be carried out based on Decree of the Minister of Health of the Republic of Indonesia Number HK.01.07/MENKES/1200/2025 concerning Guidelines for the Implementation of Cell and/or Stem Cell Therapy Services in the Field of Reconstructive and Aesthetic Plastic Surgery for various conditions such as baldness (alopecia)alopeciachronic wounds, burns, injections filler, facial rejuvenation, scar, keloid, hypertrophic scar, wound healing and regeneration, serta face contouring.
To support the implementation of these medical standards, ProSTEM provides high-quality stem cell products manufactured in facilities certified by the National Agency of Drug and Food Control (BPOM) and licensed by the Ministry of Health. ProSTEM is committed to implementing high quality and safety standards to provide safe, effective, and regulatory-compliant therapies for every patient.
References
- Crowley, J. S., Liu, A., & Dobke, M. (2021). Regenerative and stem cell-based techniques for facial rejuvenation. Experimental Biology and Medicine, 246(16), 1829–1837. https://doi.org/10.1177/15353702211020701
- Aly, R. M. (2020). Current state of stem cell-based therapies: An overview. Stem Cell Investigation, 7, 8. DOI:10.21037/sci-2020-001
- Deng, Z., Iwasaki, K., Peng, Y., & Honda, Y. (2024). Mesenchymal stem cell extract promotes skin wound healing. International Journal of Molecular Sciences, 25(24), 13745. https://doi.org/10.3390/ijms252413745
- Bojanic, C., To, K., Hatoum, A., Shea, J., Seah, K. T. M., Khan, W., & Malata, C. M. (2021). Mesenchymal stem cell therapy in hypertrophic and keloid scars. Cell and Tissue Research, 383(3), 915–930. https://doi.org/10.1007/s00441-020-03361-z
- Salibian, A. A., Widgerow, A. D., Abrouk, M., & Evans, G. R. D. (2013). Stem cells in plastic surgery: A review of current clinical and translational applications. Archives of Plastic Surgery, 40(6), 666–675. DOI:10.5999/aps.2013.40.6.666