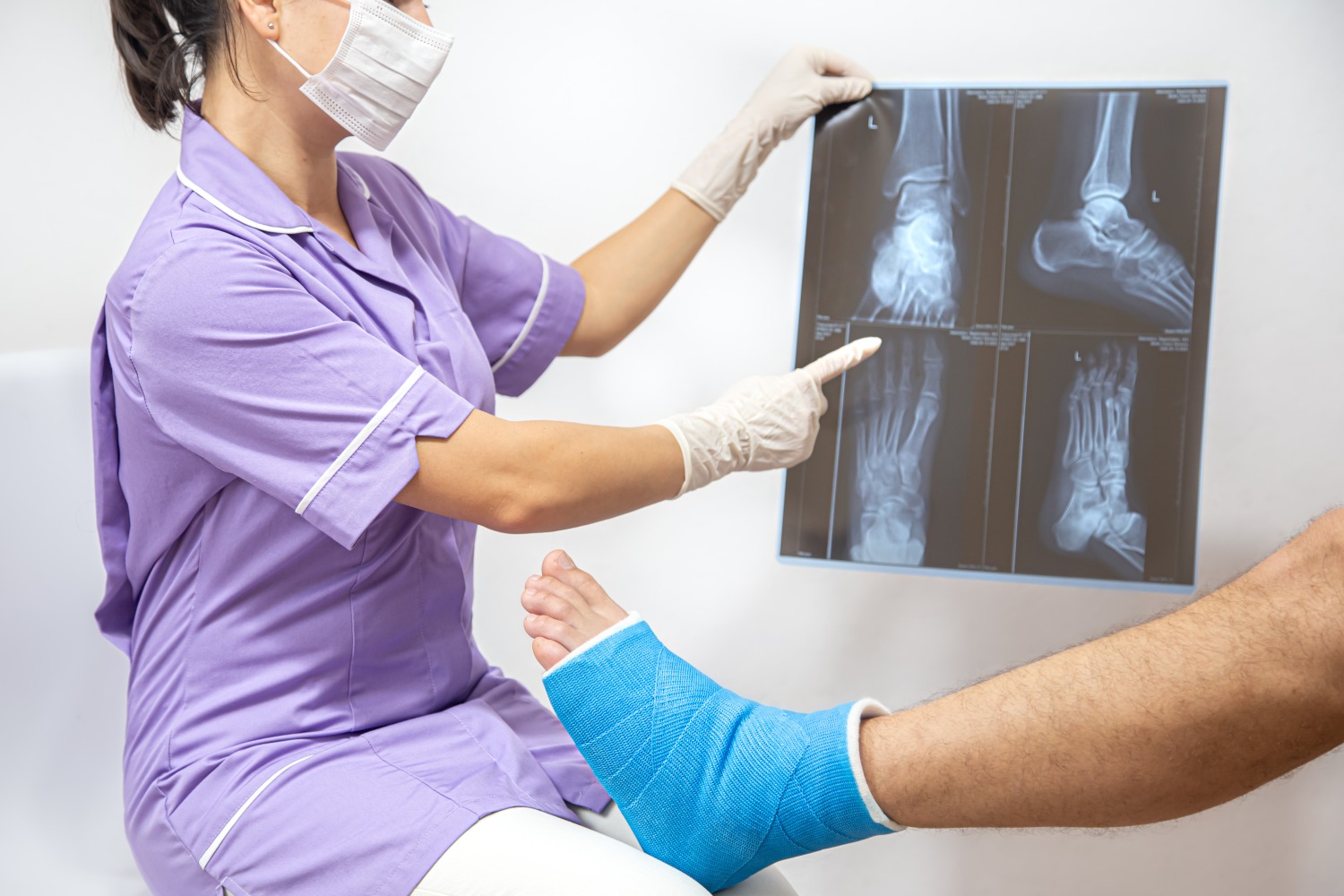
A non-union fracture is a condition in which the broken bone fails to unite or heal properly within the expected healing time. Based on the definition of Food and Drug Administration USA, non-union is a condition where a broken bone has not shown any signs of healing after nine months after the injury, and there has been no progress in healing for three consecutive months, thus potentially causing further complications. [1].
The severity of this condition is assessed based on its impact on movement function, physical changes, treatment difficulties, and the patient's risk factors. This makes it difficult to generalize the outcome of non-union fractures across all patients. Factors that significantly contribute to the condition include obesity, diabetes, and smoking. [2]. To manage and treat this condition, a combination of medical procedures is performed to ensure the bone structure remains solid (does not shift), along with a series of therapies that can help new bone grow better. In cases of infected bones, treatment may involve a frame attached to the outside of the bone to prevent the infection from spreading to other parts. [3].
Currently, stem cell therapy is gaining attention due to its ability to transform into new bone cells. This ability enhances bone repair by promoting overall healing. For example, stem cell therapy uses mesenchymal stem cells. Umbilical cord stem cell (MSC) and stem cell-derived secretome have shown promising results in enhancing healing in non-healing bones. Although this treatment method has not yet been standardized and requires further testing, the potential for stem cell therapy in the future appears promising. [4,5].
Now, stem cell therapy services for orthopedic cases, especially non-union fractures, can be carried out based on the Decree of the Minister of Health of the Republic of Indonesia (KMK) number HK.01.07/MENKES/1359/2024 concerning Guidelines for the Implementation of Stem Cell Therapy Services in the Field of Orthopedics and Traumatology.
ProSTEM is here to provide stem cells and secretomes Produced in a facility licensed by the Food and Drug Authority (BPOM) and the Ministry of Health, ProSTEM demonstrates our dedication to quality and safety standards. We ensure that every therapeutic service we provide is safe, effective, and complies with all medical regulations and government requirements for patient safety. For more information about stem cell therapy for non-union fractures, please contact us via WhatsApp on this website.
References
- Thomas, J. D., & Kehoe, J. L. (2023). Bone nonunion. StatPearls – NCBI Bookshelf. https://www.ncbi.nlm.nih.gov/books/NBK554385/
- Ghanem, W., Ezzeddine, H., Saad, R., Kiwan, E., Dahdouh, R., Fakih, O., Sakhat, G., Alam, E., Najjar, J., Assaf, F., Chahine, M., Dib, N., Kortbawi, R., Badra, M., & Moucharafieh, R. (2025). State of the Nonunion: A review of the latest literature. Orthopedic Reviews, 17, 129085. https://doi.org/10.52965/001c.129085
- Nicholson, J., Makaram, N., Simpson, A., & Keating, J. (2020). Fracture nonunion in long bones: A literature review of risk factors and surgical management. Injury, 52, S3–S11. https://doi.org/10.1016/j.injury.2020.11.029
- Smolinska, V., Csobonyeiova, M., Zamborsky, R., & Danisovic, L. (2023). Stem cells and their derivatives: an implication for the regeneration of nonunion fractures. Cell Transplantation, 32, 9636897231183530. https://doi.org/10.1177/09636897231183530
- Dilogo, I. H., Fiolin, J., Canintika, A. F., Pawitan, J. A., & Luviah, E. (2022). The effect of secretome, xenogenic bone Marrow-Derived mesenchymal stem cells, bone morphogenetic protein-2, hydroxyapatite granule and mechanical fixation in Critical-Size defects of RAT models. pmc.ncbi.nlm.nih.gov. https://doi.org/10.22038/ABJS.2021.49539.2458