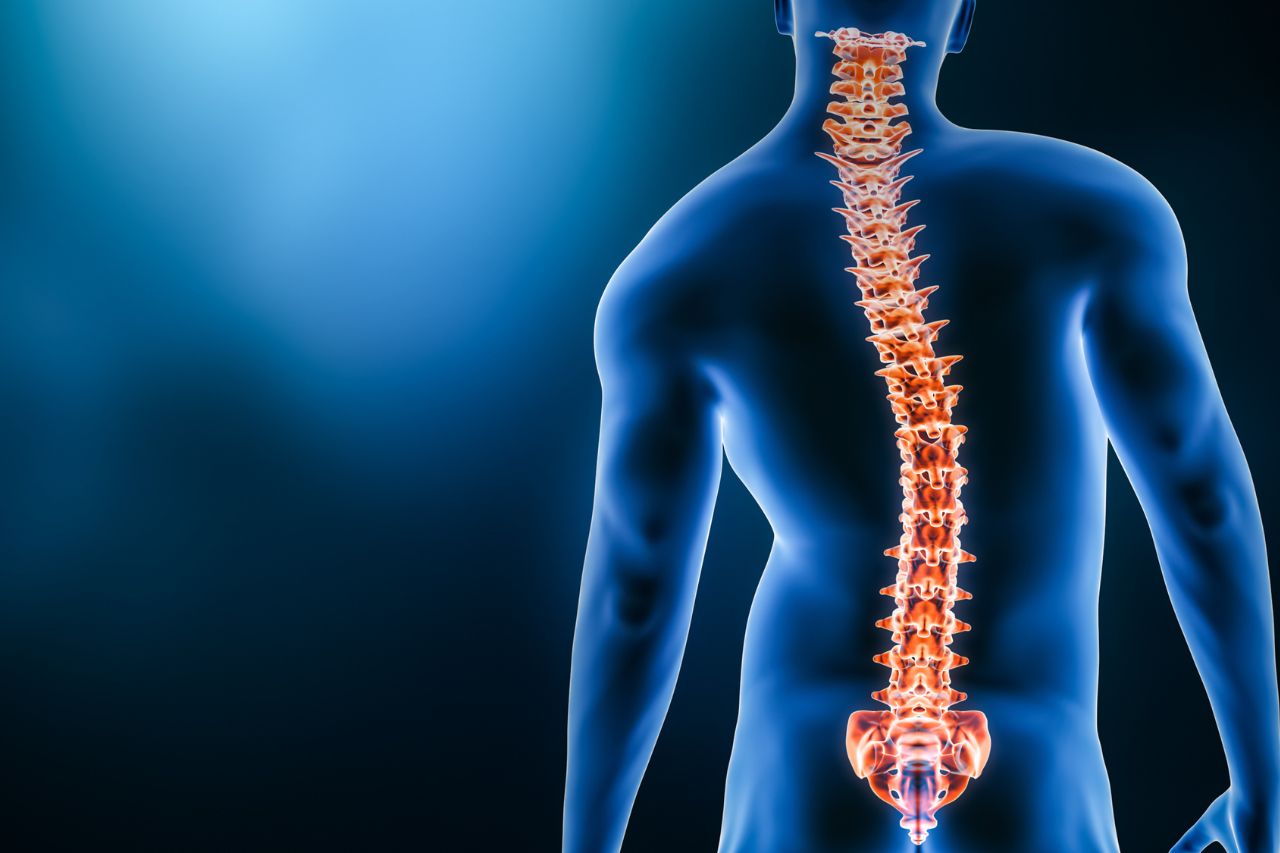
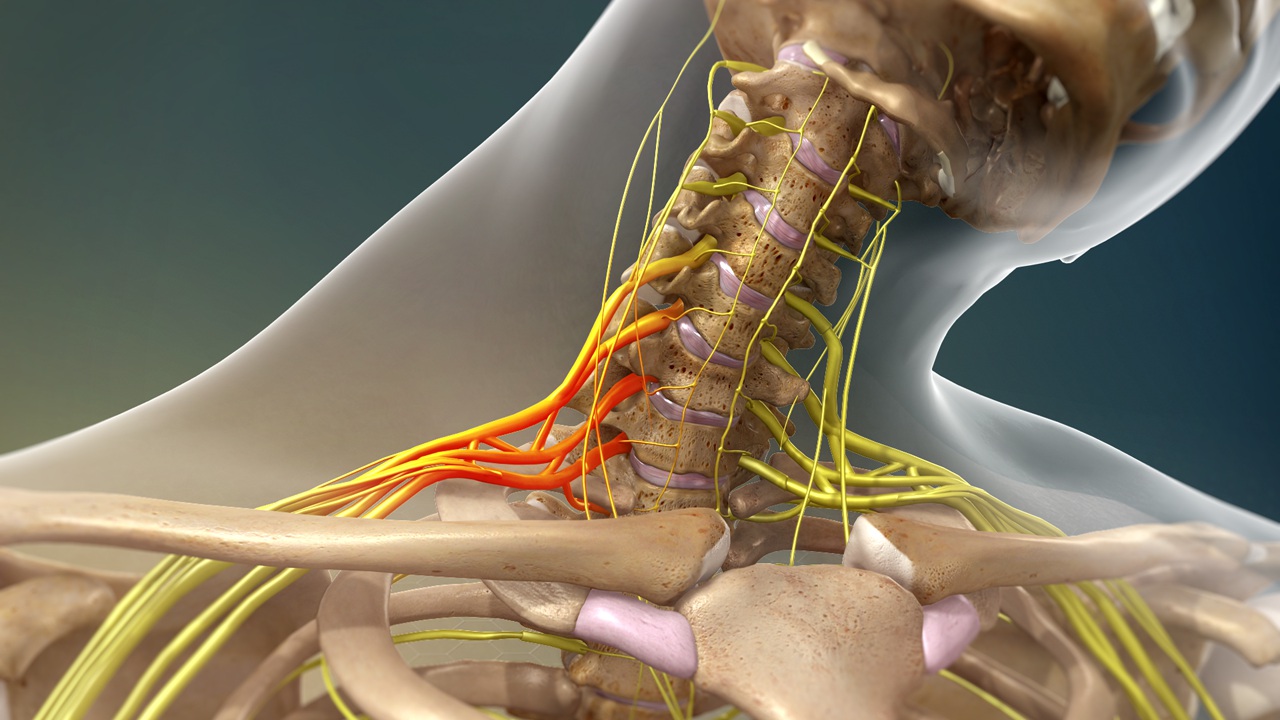
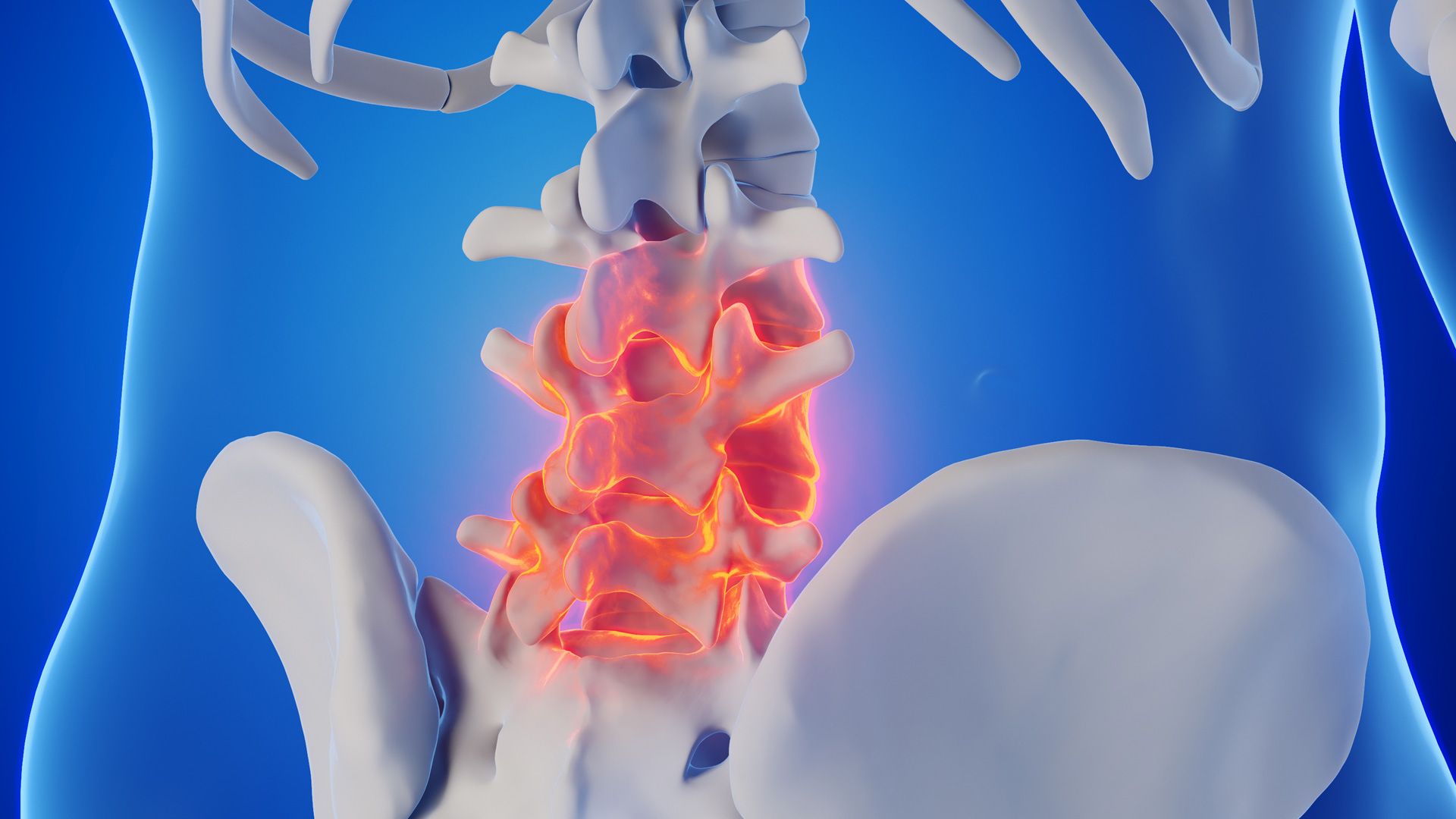
Congenital scoliosis, including conditions hermivertebra, is a congenital abnormality of the spine that occurs due to disruption of vertebral formation during embryonic development. In this condition hemivertebra, One side of the spinal column does not form properly, causing the spine to curve in an "S" or "C" shape. This abnormality occurs during pregnancy, particularly in the first trimester, when the spine is forming. The cause is generally related to impaired embryonic development and is not directly influenced by the mother's diet or lifestyle during pregnancy. In some cases, this condition may also be related to genetic factors.. Common symptoms include shoulders that appear misaligned, a waist or pelvis that tilts to one side, and a bulge in the back when bending over, reflecting overall postural asymmetry.[1].
The severity of congenital scoliosis can vary and can potentially progress as the child grows. Therefore, management is individualized, ranging from regular observation and brace use to surgery in certain cases. Comprehensive structural correction remains a challenge in treating this condition.[2]
In treating this condition, stem cell therapy using mesenchymal stem cells from umbilical cord shows promise as a promising, minimally invasive therapy for correcting existing curvatures and preventing future spinal curvatures. This healing mechanism is driven by the ability of stem cells to transform into new bone cells that can become the building blocks of the spine. [3]. In addition, stem cell therapy that uses processed stem cells, namely secretome tengah diteliti terkait potensinya pada kondisi kelainan dan penyakit neonatal, such as congenital scoliosis lesions. In theory, secretome contains biological molecules that can help reduce inflammation and promote healing in the body[4]However, the application of this therapy is still under testing and requires further adjustment and standardization for use in humans.
However, the application of this therapy is still under testing and requires further adaptation and standardization for use in humans.
ProSTEM provides high-quality stem cell and secretome products manufactured in facilities certified by the National Agency of Drug and Food Control (BPOM) and licensed by the Ministry of Health. This commitment reflects our commitment to maintaining quality, safety, and compliance with applicable regulations in every service we provide. For more information about stem cell-based therapies, please contact our official service provider through the provided contact information.
References
- Grabala, P. (2025). Congenital Scoliosis: A Comprehensive Review of Diagnosis, Management, and Surgical Decision-Making in Pediatric Spinal Deformity—An Expanded Narrative Review. Journal of Clinical Medicine, 14(22), 8085. https://doi.org/10.3390/jcm14228085
- Peng Z., Zhang H., Wang S., Zhang J. (2025). Advances and the Diagnosis and Treatment of Congenital Scoliosis. European Journal of Medical Research 30:683
- Rahyussalim, A. J., Nasser, M. K., As’ady, F. M. A., & Kurniawati, T. (2022). Umbilical cord-derived mesenchymal stem cells implantation on Hemivertebra defect with three-year follow-up: Biological approach in congenital scoliosis treatment – A case report. International Journal of Surgery Case Reports, 99, 107602. https://doi.org/10.1016/j.ijscr.2022.107602
- Tung, S., Delavogia, E., Fernandez-Gonzalez, A., Mitsialis, S. A., & Kourembanas, S. (2023). Harnessing the therapeutic potential of the stem cell secretome in neonatal diseases. Seminars in Perinatology, 47(3), 151730. https://doi.org/10.1016/j.semperi.2023.151730